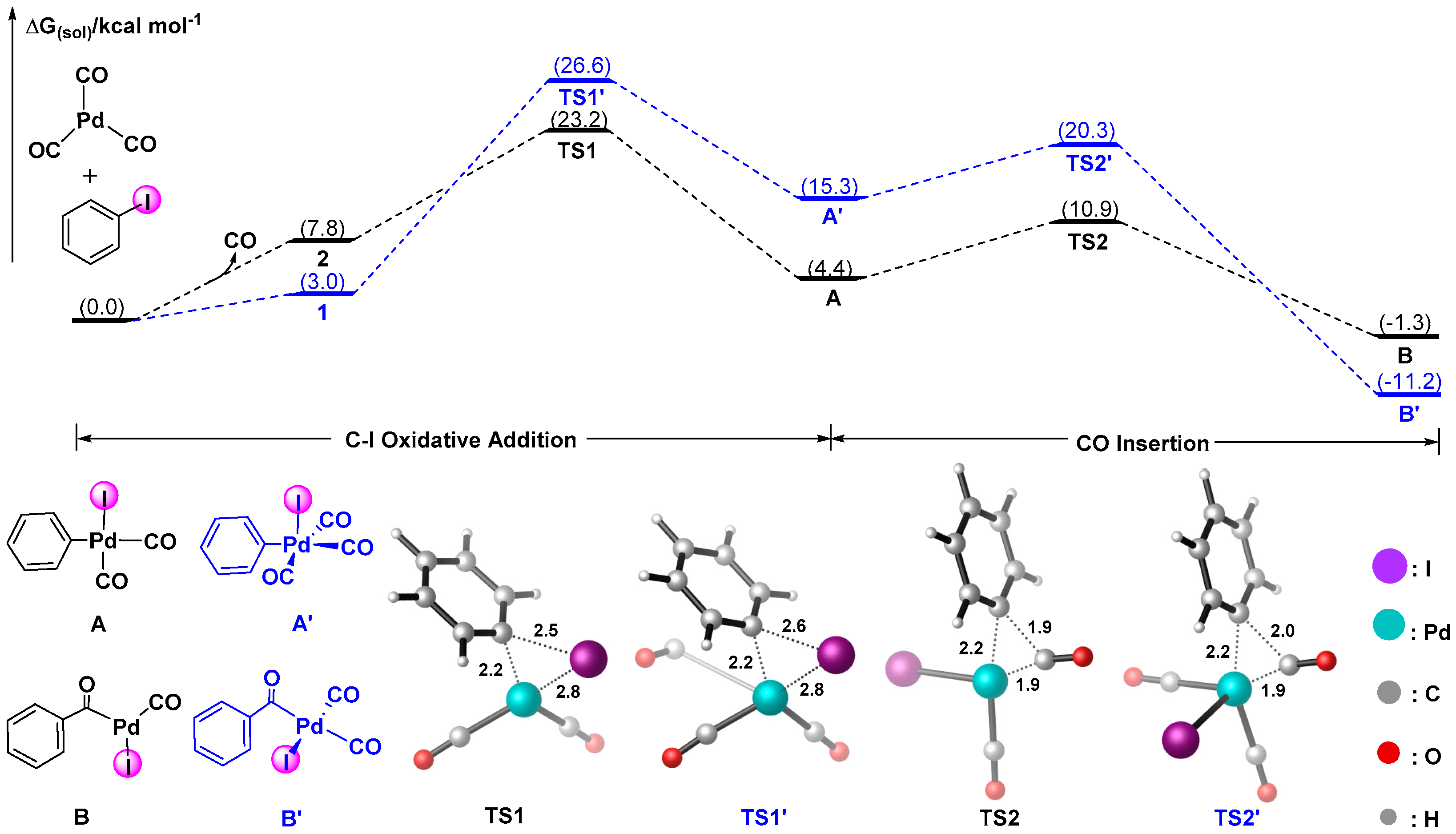
Catalysts | Free Full-Text | A Theoretical Study on Pd-catalyzed, Friedel-Crafts Intermolecular Acylation: Does Generated In Situ Aroyl Triflate Act as A Reactive Electrophile to Functionalize C–H Bond of Arenes? | HTML

Nucleophilic palladium-catalysed Ar−F bond-forming reaction. a, The... | Download Scientific Diagram

Palladium‐Catalyzed Elimination/Isomerization of Enol Triflates into 1,3‐Dienes - Crouch - 2011 - Angewandte Chemie International Edition - Wiley Online Library
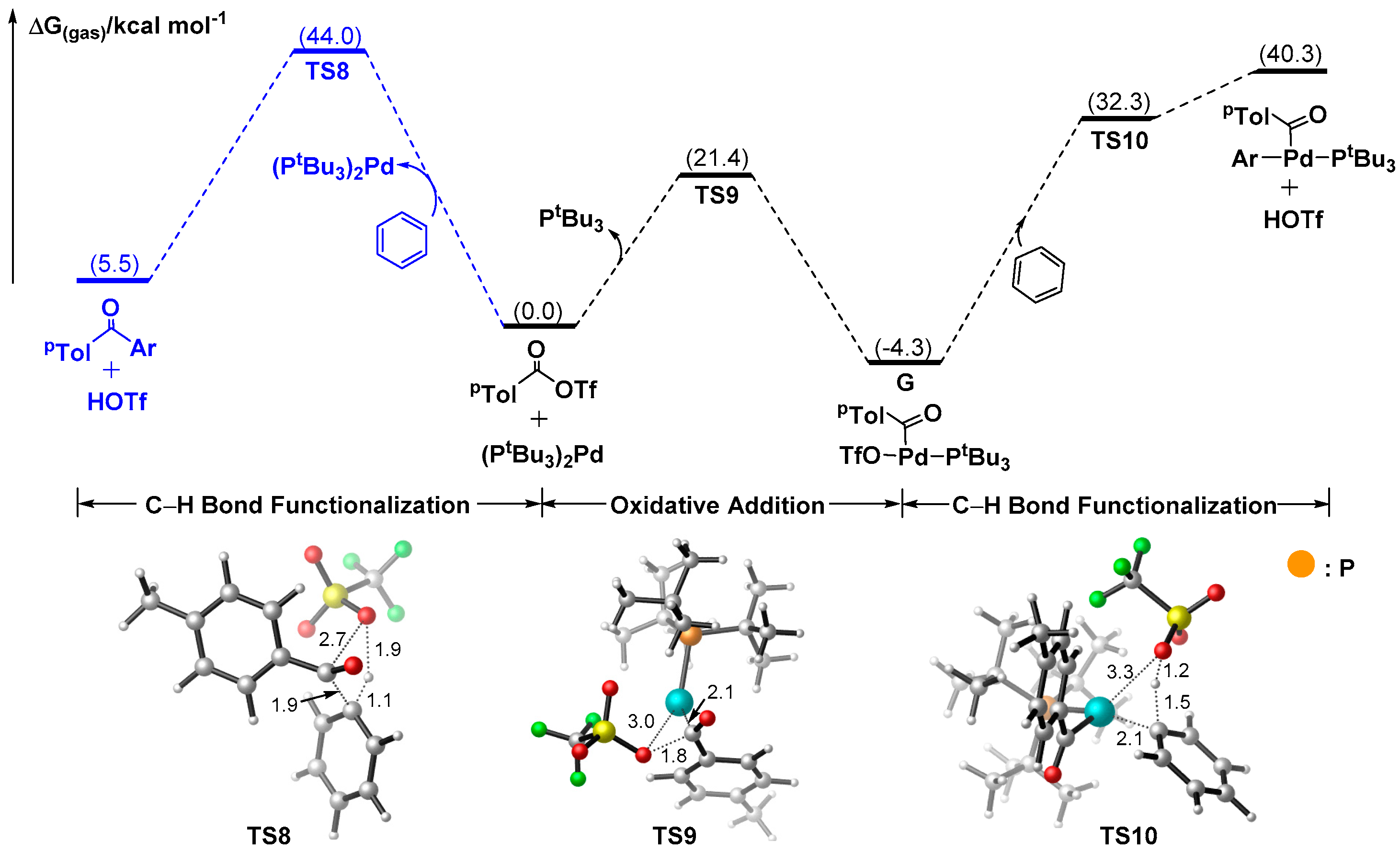
Catalysts | Free Full-Text | A Theoretical Study on Pd-catalyzed, Friedel-Crafts Intermolecular Acylation: Does Generated In Situ Aroyl Triflate Act as A Reactive Electrophile to Functionalize C–H Bond of Arenes? | HTML

Recent Progress in the Use of Pd-Catalyzed C-C Cross-Coupling Reactions in the Synthesis of Pharmaceutical Compounds
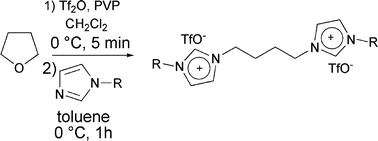
Halide-free highly-pure imidazolium triflate ionic liquids: Preparation and use in palladium-catalysed allylic alkylation - Green Chemistry (RSC Publishing)
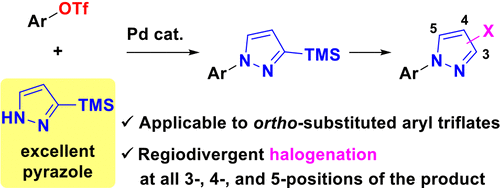
Synthesis of N-Arylpyrazoles by Palladium-Catalyzed Coupling of Aryl Triflates with Pyrazole Derivatives - J. Org. Chem. - X-MOL

Mild Palladium-Catalyzed Cyanation of (Hetero)aryl Halides and Triflates in Aqueous Media- | Sigma-Aldrich
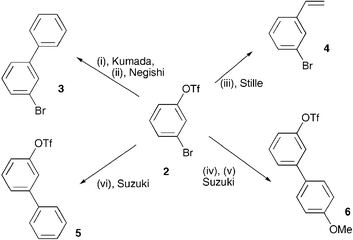
Aryl bromide/triflate selectivities reveal mechanistic divergence in palladium-catalysed couplings; the Suzuki–Miyaura anomaly - Chemical Communications (RSC Publishing) DOI:10.1039/B701517H

Palladium-catalyzed triethylammonium formate reduction of aryl triflates. A selective method for the deoxygenation of phenols - ScienceDirect

Palladium-catalysed dimerization of vinylarenes using indium triflate as an effective co-catalyst - Chemical Communications (RSC Publishing)
![R)-(+)-2,2′-Bis(diphenylphosphino)-1,1′-binaphthyl]-diaquo-palladium(II) bis(triflate) | Sigma-Aldrich R)-(+)-2,2′-Bis(diphenylphosphino)-1,1′-binaphthyl]-diaquo-palladium(II) bis(triflate) | Sigma-Aldrich](https://www.sigmaaldrich.com/content/dam/sigma-aldrich/structure3/041/a_____705780.eps/_jcr_content/renditions/a_____705780-medium.png)







![1,3-Bis(diphenylphosphino)propane]palladium(II) triflate | 137846-38-9 | Sigma-Aldrich 1,3-Bis(diphenylphosphino)propane]palladium(II) triflate | 137846-38-9 | Sigma-Aldrich](https://www.sigmaaldrich.com/content/dam/sigma-aldrich/structure8/037/a_____705772.eps/_jcr_content/renditions/a_____705772-medium.png)