![Bis[(diphenylphosphanyl)methyl]amine palladium(II) acetate, polymer-bound | Sigma-Aldrich | Sigma-Aldrich Bis[(diphenylphosphanyl)methyl]amine palladium(II) acetate, polymer-bound | Sigma-Aldrich | Sigma-Aldrich](https://www.sigmaaldrich.com/content/dam/sigma-aldrich/structure4/102/mfcd07370614.eps/_jcr_content/renditions/mfcd07370614-medium.png)
Bis[(diphenylphosphanyl)methyl]amine palladium(II) acetate, polymer-bound | Sigma-Aldrich | Sigma-Aldrich
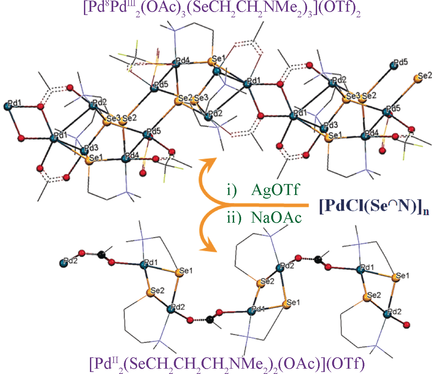
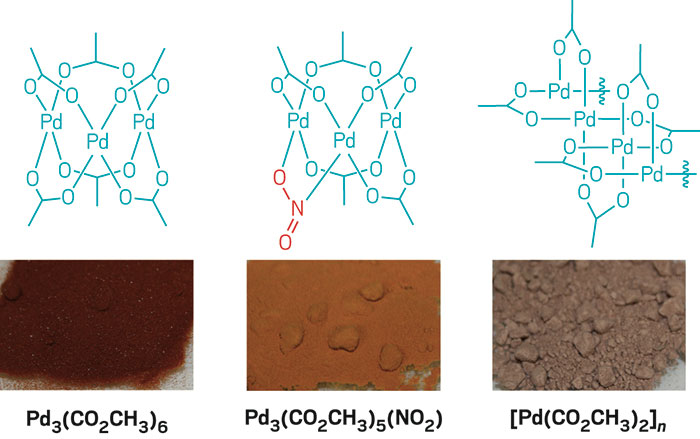
Coordination Polymers of Palladium Bridged by Carboxylate and Dimethylaminoalkylselenolate Ligands - Chem. Asian J. - X-MOL
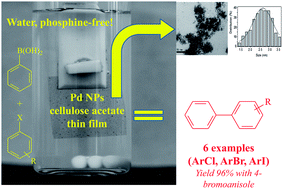
Palladium nanoparticles supported in a polymeric membrane: an efficient phosphine-free “green” catalyst for Suzuki–Miyaura reactions in water - RSC Advances (RSC Publishing)

PDF) Polymer Supported Triphenylphosphine-Palladium Acetate Complex PS-TPP- Pd(OAc)(2) as a Heterogeneous and Reusable Catalyst for Indirect Reductive Amination of Aldehydes
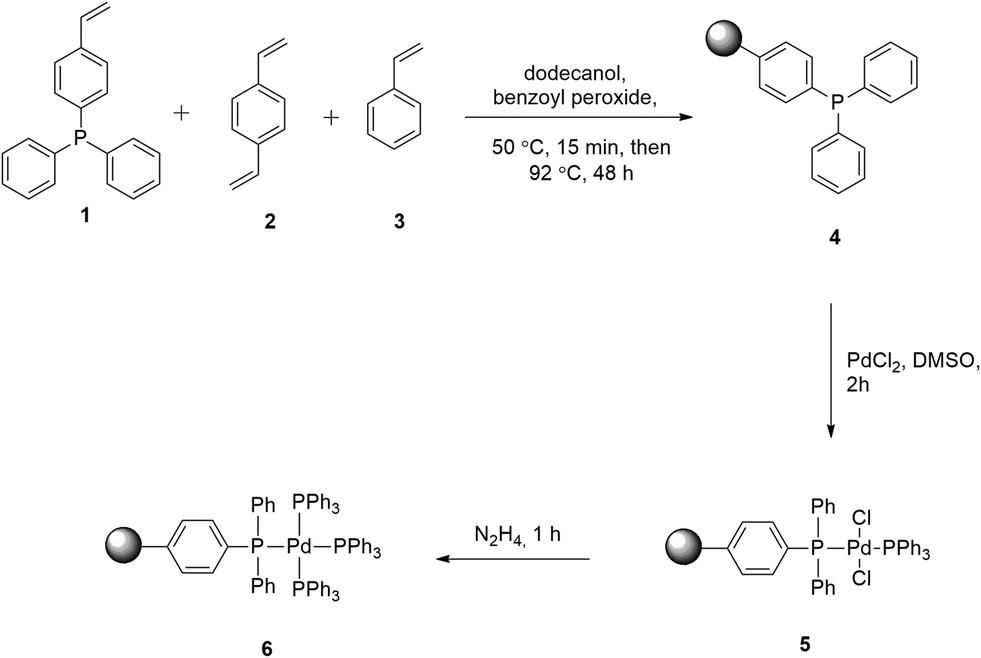
Immobilized tetrakis(triphenylphosphine)palladium(0) for Suzuki–Miyaura coupling reactions under flow conditions - Reaction Chemistry & Engineering (RSC Publishing)

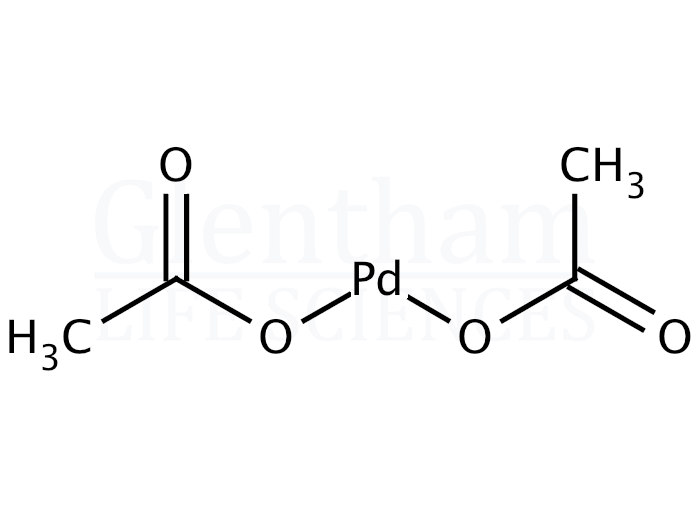
Understanding Palladium Acetate from a User Perspective - Carole - 2016 - Chemistry – A European Journal - Wiley Online Library
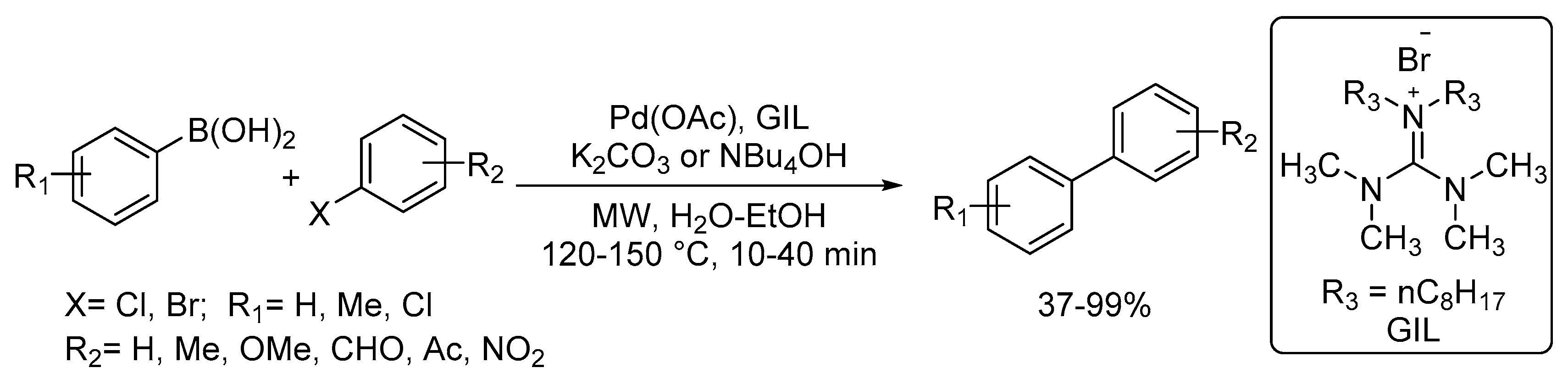
Catalysts | Free Full-Text | Microwave-Assisted Palladium-Catalyzed Cross-Coupling Reactions: Generation of Carbon–Carbon Bond | HTML
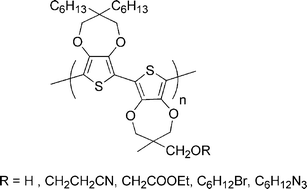
Single step reductive polymerization of functional 3,4-propylenedioxythiophenes via direct C–H arylation catalyzed by palladium acetate - Polymer Chemistry (RSC Publishing)

Di(acetato)dicyclohexylphenylphosphinepalladium(II), polymer-bound FibreCat® Pd ~ 5 %, loading 0.4-0.6mmol/g PPH3 ligand content | 480445-43-0 | Sigma-Aldrich

Palladium acetate supported on amidoxime-functionalized magnetic cellulose: Synthesis, DFT study and application in Suzuki reaction - ScienceDirect
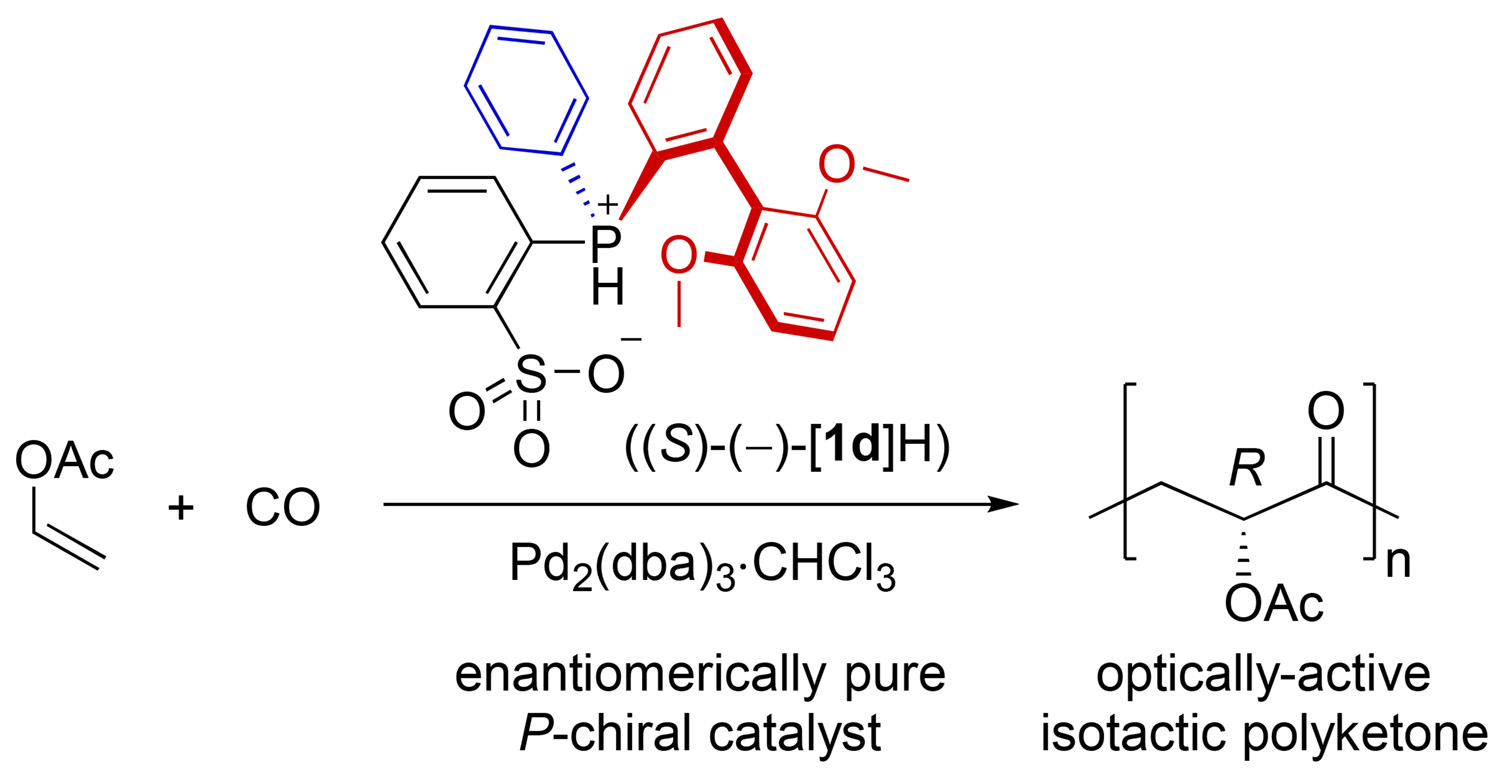
P-Chiral Phosphine–Sulfonate/Palladium-Catalyzed Asymmetric Copolymerization of Vinyl Acetate with Carbon Monoxide - Ito Research Group

Palladium Nanoparticles within Core-Cross-Linked Polymer Gels for Suzuki Coupling Reactions: from Monomers to Ready-to-Use Catalysts in Two-Steps

Nanosized palladium-catalyzed Suzuki–Miyaura coupling polymerization: synthesis of soluble aromatic poly(ether ketone)s | Polymer Journal
![3375-31-3・Palladium(Ⅱ) Acetate・165-24701・161-24703・163-24702[Detail Information] | [Synthesis & Materials] |Laboratory Chemicals-FUJIFILM Wako Chemicals U.S.A. Corporation 3375-31-3・Palladium(Ⅱ) Acetate・165-24701・161-24703・163-24702[Detail Information] | [Synthesis & Materials] |Laboratory Chemicals-FUJIFILM Wako Chemicals U.S.A. Corporation](https://labchem-wako.fujifilm.com/sc/01/3375-31-3.png)
3375-31-3・Palladium(Ⅱ) Acetate・165-24701・161-24703・163-24702[Detail Information] | [Synthesis & Materials] |Laboratory Chemicals-FUJIFILM Wako Chemicals U.S.A. Corporation

A) Palladium/benzoic acid-catalyzed polymerization of internal diynes... | Download Scientific Diagram

Palladium Nanoparticles within Core-Cross-Linked Polymer Gels for Suzuki Coupling Reactions: from Monomers to Ready-to-Use Catalysts in Two-Steps
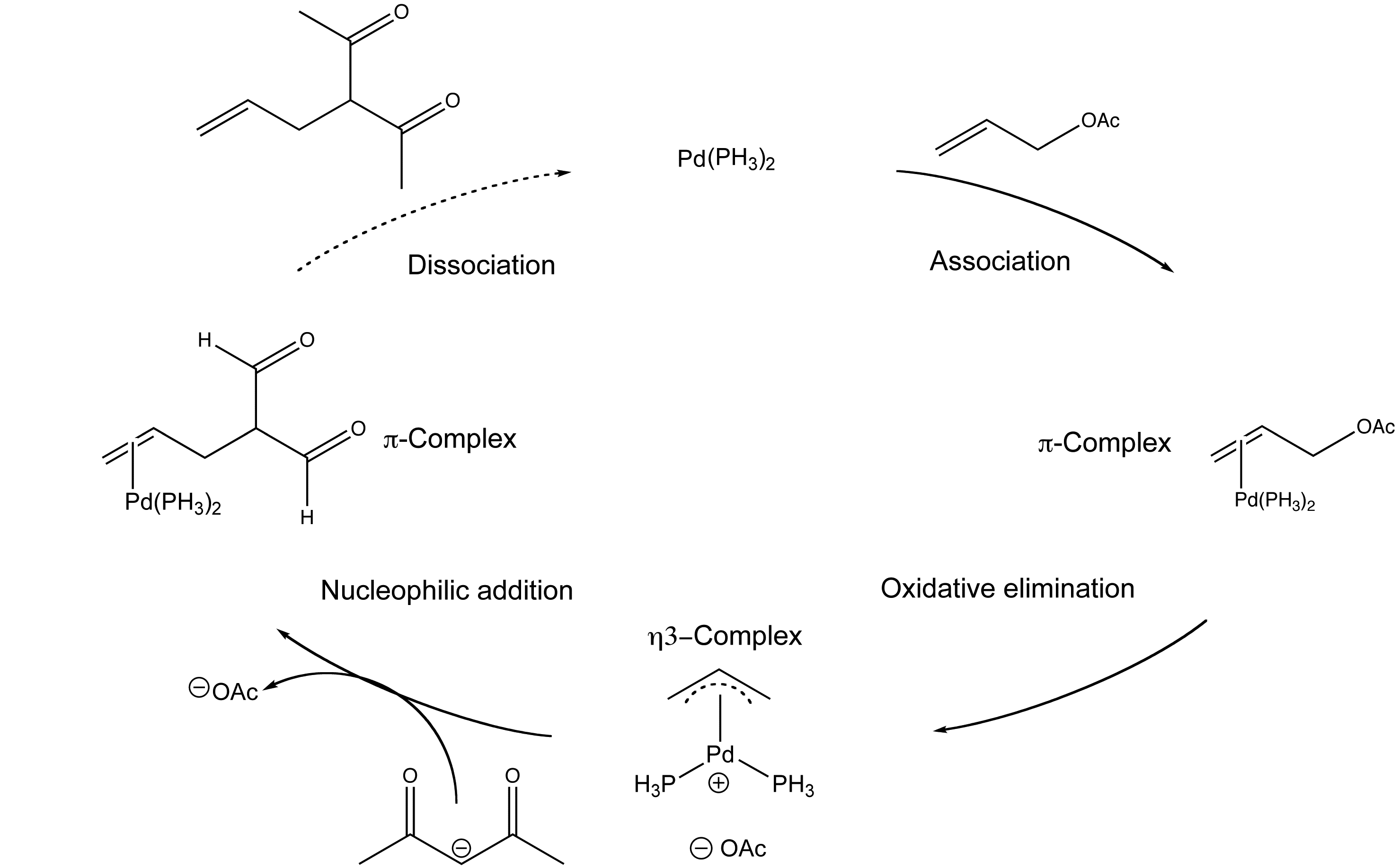
Organopalladium Chemistry - Palladium-catalysed nucleophilic allylic substitution of functionalised compounds
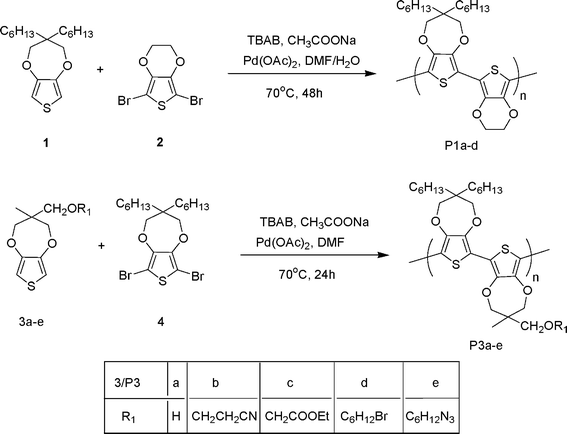
Single step reductive polymerization of functional 3,4-propylenedioxythiophenes via direct C–H arylation catalyzed by palladium acetate - Polymer Chemistry (RSC Publishing) DOI:10.1039/B9PY00265K
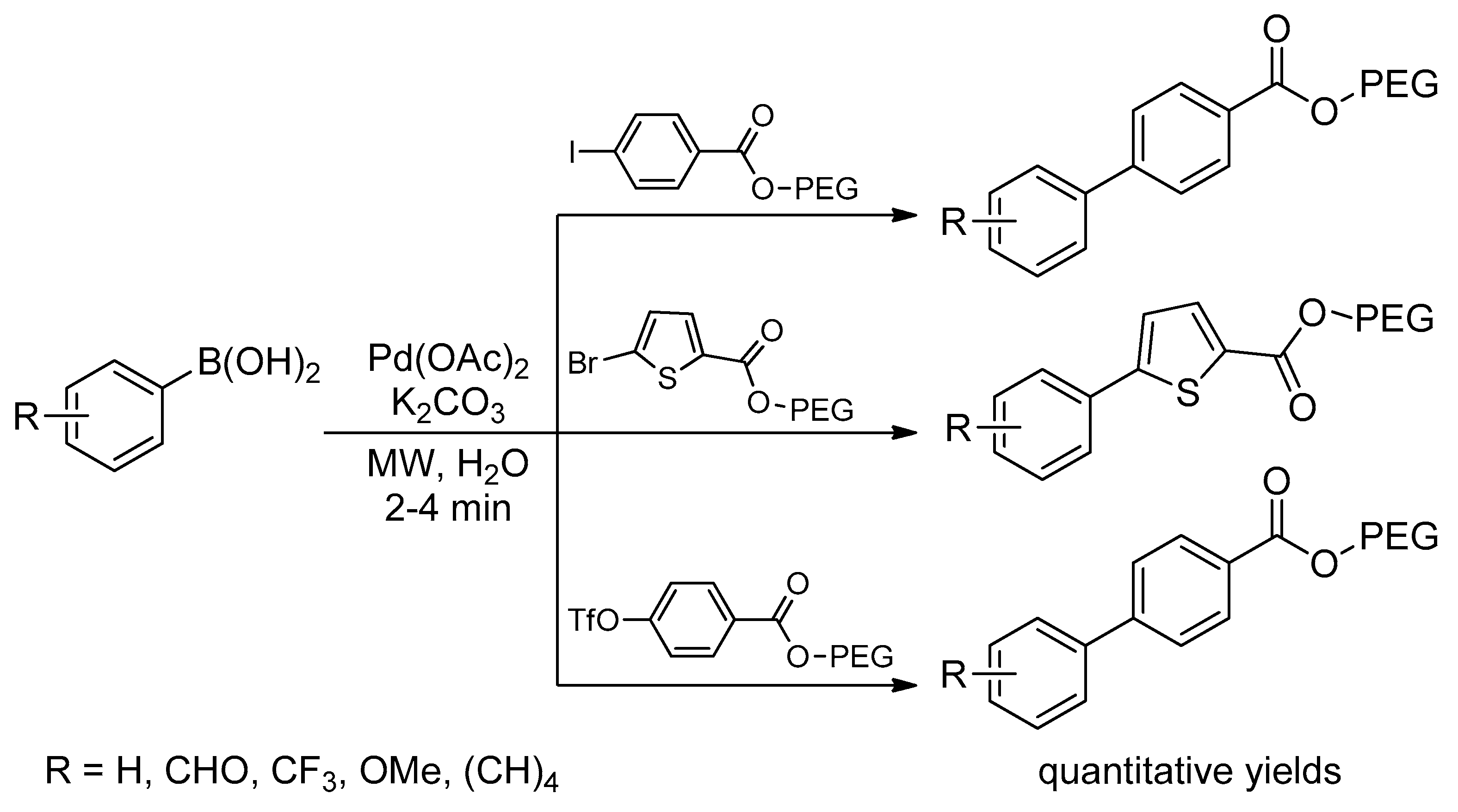
Catalysts | Free Full-Text | Microwave-Assisted Palladium-Catalyzed Cross-Coupling Reactions: Generation of Carbon–Carbon Bond | HTML
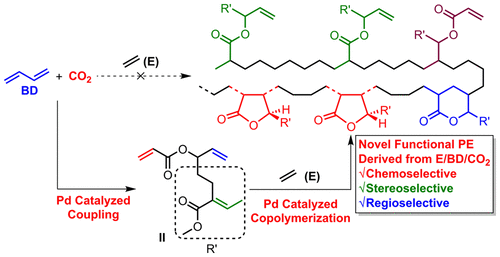
![Bis[(diphenylphosphanyl)methyl]amine palladium(II) acetate, polymer -bound|Sigma... Bis[(diphenylphosphanyl)methyl]amine palladium(II) acetate, polymer -bound|Sigma...](http://en.chembase.cn/Server/MolImages/e4/a0/e4a01ff2-e5ee-45f5-85d3-915ceb54359f.png)