
Palladium-Catalyzed Intramolecular 5-exo-dig Hydroarylations of N-Arylpropiolamides: Thermodynamics-Controlled Stereoselective Synthesis of 3-Methyleneoxindoles | The Journal of Organic Chemistry

N, O-polydentate ligands for palladium-catalyzed cross-coupling reactions (Part III) - ScienceDirect
![Optimization of Levulinic Acid Production from Depithed Sugarcane Bagasse in 1- Ethyl-3-methylimidazolium hydrogen sulfate [EMim][HSO4] | SpringerLink Optimization of Levulinic Acid Production from Depithed Sugarcane Bagasse in 1- Ethyl-3-methylimidazolium hydrogen sulfate [EMim][HSO4] | SpringerLink](https://media.springernature.com/lw685/springer-static/image/art%3A10.1007%2Fs12649-020-01221-z/MediaObjects/12649_2020_1221_Figa_HTML.png)
Optimization of Levulinic Acid Production from Depithed Sugarcane Bagasse in 1- Ethyl-3-methylimidazolium hydrogen sulfate [EMim][HSO4] | SpringerLink
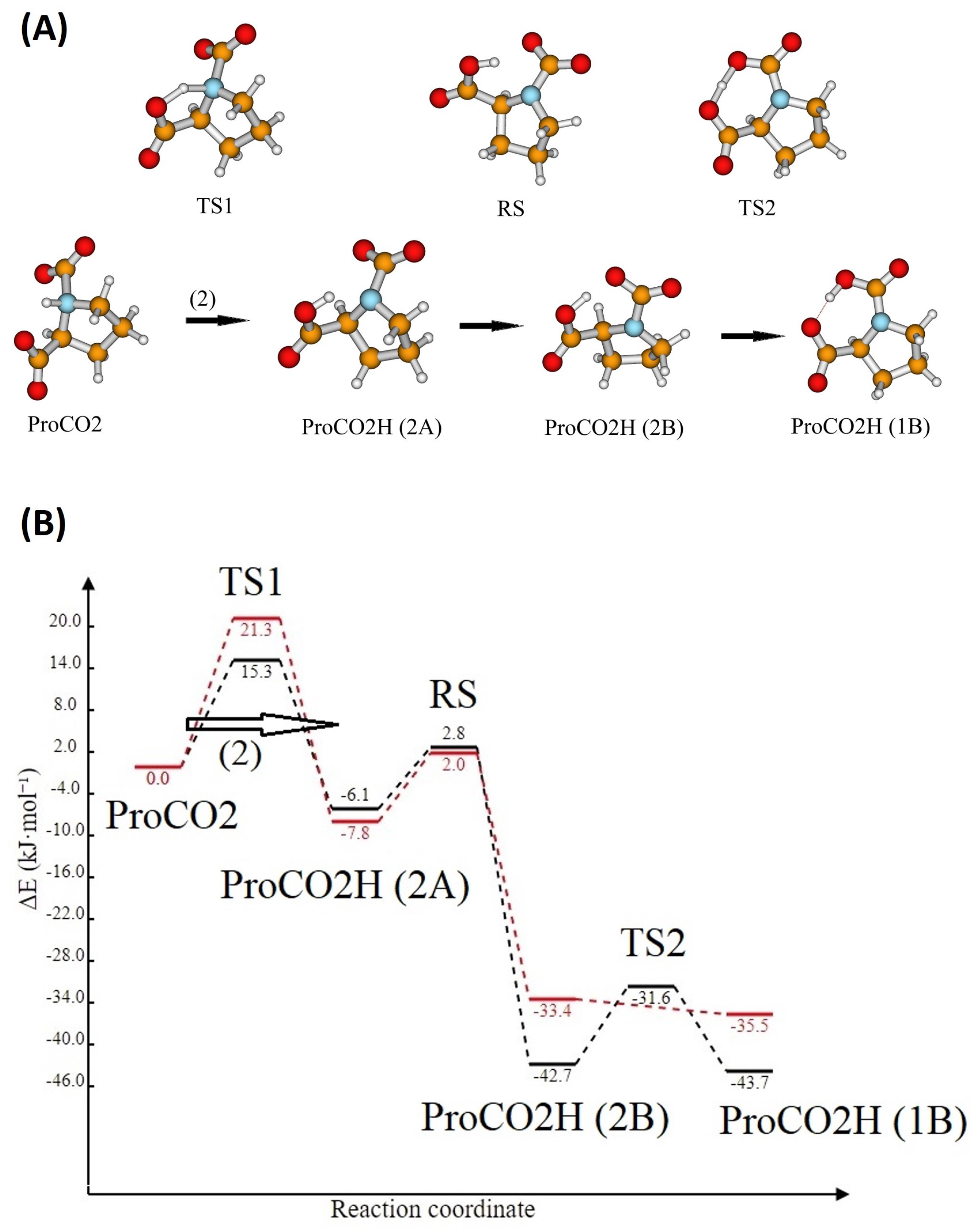
Entropy | Free Full-Text | Reaction Mechanism of CO2 with Choline-Amino Acid Ionic Liquids: A Computational Study

1,3-Bis(carboxymethyl)imidazolium Chloride as a Metal-Free and Recyclable Catalyst for the Synthesis of N-Allylanilines by Allylic Substitution of Alcohols | ACS Sustainable Chemistry & Engineering
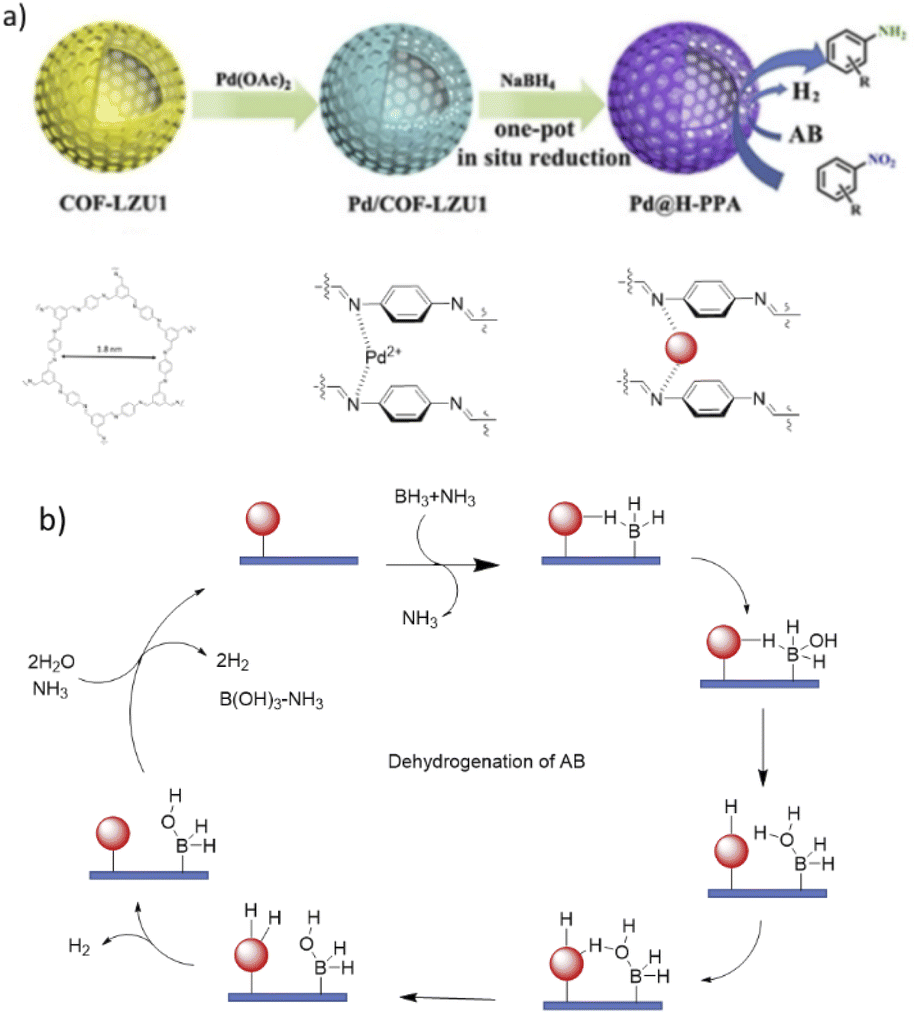
Covalent organic framework supported palladium catalysts - Journal of Materials Chemistry A (RSC Publishing) DOI:10.1039/D2TA05234B

Mild Palladium-Catalyzed Selective Monoarylation of Nitriles | Journal of the American Chemical Society

Synthesis of Benzothiophene-3-carboxylic Esters by Palladium Iodide-Catalyzed Oxidative Cyclization–Deprotection–Alkoxycarbonylation Sequence under Aerobic Conditions - ScienceDirect

Structures of acridine derivatives examined for effect on TDP-43 2C... | Download Scientific Diagram

Trimetallic PEPPSI‐Type Palladium N‐Heterocyclic Carbene Complexes – Improved Catalyst Lifetime in the Mizoroki–Heck Coupling Reaction - Martínez‐Olid - 2015 - European Journal of Inorganic Chemistry - Wiley Online Library

Nonsolvent application of ionic liquids: organo-catalysis by 1-alkyl-3- methylimidazolium cation based room-temperature ionic liquids for chemoselective N-tert-butyloxycarbonylation of amines and the influence of the C-2 hydrogen on catalytic efficiency ...
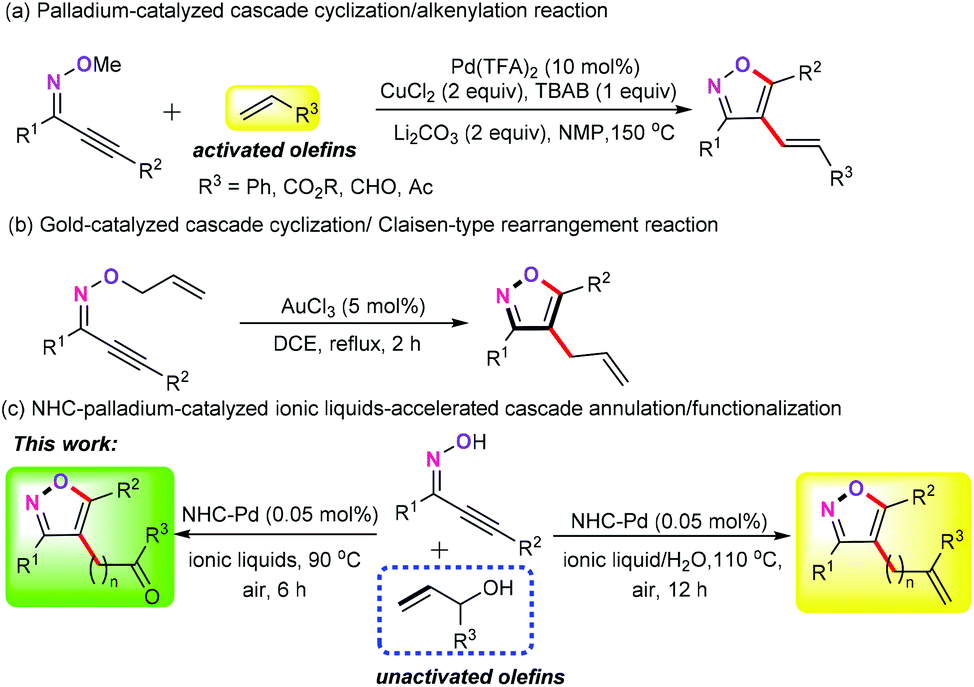
Palladium-catalyzed ionic liquid-accelerated oxidative annulation of acetylenic oximes with unactivated long-chain enols - Green Chemistry (RSC Publishing) DOI:10.1039/D0GC02037K







![Crystal structure of bis(1-ethyl-3-methylimidazolium) tetrabromidocadmate(II), [C6H11N2]2[CdBr4] Crystal structure of bis(1-ethyl-3-methylimidazolium) tetrabromidocadmate(II), [C6H11N2]2[CdBr4]](https://www.degruyter.com/document/doi/10.1515/ncrs-2015-0117/asset/graphic/j_ncrs-2015-0117_fx_1.jpg)
