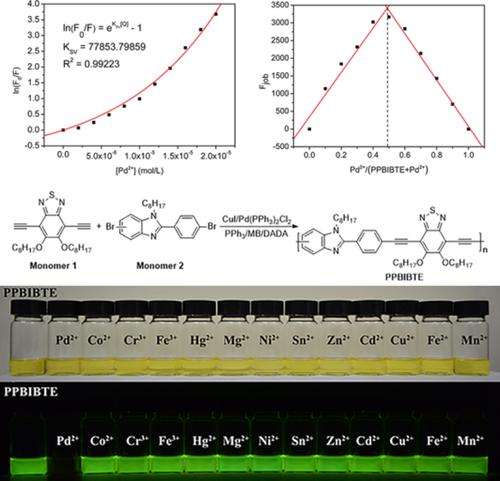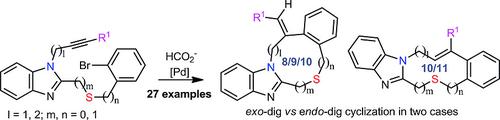
Synthesis of Benzimidazole‐Fused Medium‐Sized N,S‐Heterocycles via Palladium‐Catalyzed Cyclizations,European Journal of Organic Chemistry - X-MOL

Arylation of heterocyclic compounds by benzimidazole-based N-heterocyclic carbene-palladium(II) complexes - ScienceDirect

Enhanced separation of Pd(II) and Pt(IV) from hydrochloric acid aqueous solution using 2-((2-methoxyethyl)thio)-1H-benzimidazole | SpringerLink
Heck reaction using palladium-benzimidazole catalyst: synthesis, characterisation and catalytic activity
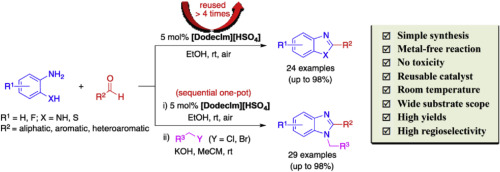
Metal-free selective synthesis of 2-substituted benzimidazoles catalyzed by Brönsted acidic ionic liquid: Convenient access to one-pot synthesis of N-alkylated 1,2-disubstituted benzimidazoles - Tetrahedron - X-MOL

Arylation of heterocyclic compounds by benzimidazole-based N-heterocyclic carbene-palladium(II) complexes - ScienceDirect

Sequential Metal‐Catalyzed N‐Heteroarylation and C–C Cross‐Coupling Reactions: An Expedient Route to Tris(hetero)aryl Systems - Siddle - 2008 - European Journal of Organic Chemistry - Wiley Online Library

PDF) An Efficient Synthesis of Benzimidazoles via Palladium-Catalyzed Amine Exchange Reaction from Trialkylamines to o-Phenylenediamine in an Aqueous Medium

Scheme 1 | Catalytic activity of a supported palladium–benzimidazole complex toward alkene hydrogenation | SpringerLink

Ionically tagged benzimidazole palladium(II) complex: preparation and catalytic application in cross-coupling reactions - ScienceDirect
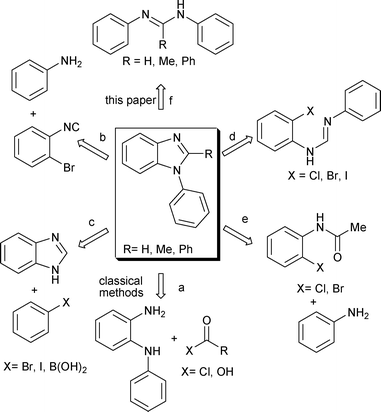
Palladium -catalyzed aerobic oxidative C–H amination: synthesis of 2-unsubstituted and 2-substituted N -aryl benzimidazoles - RSC Advances (RSC Publishing) DOI:10.1039/C2RA20328F

Benzimidazole bearing Pd–PEPPSI complexes catalyzed direct C2‐arylation/heteroarylation of N‐substituted benzimidazoles - Gokanapalli - 2020 - Applied Organometallic Chemistry - Wiley Online Library
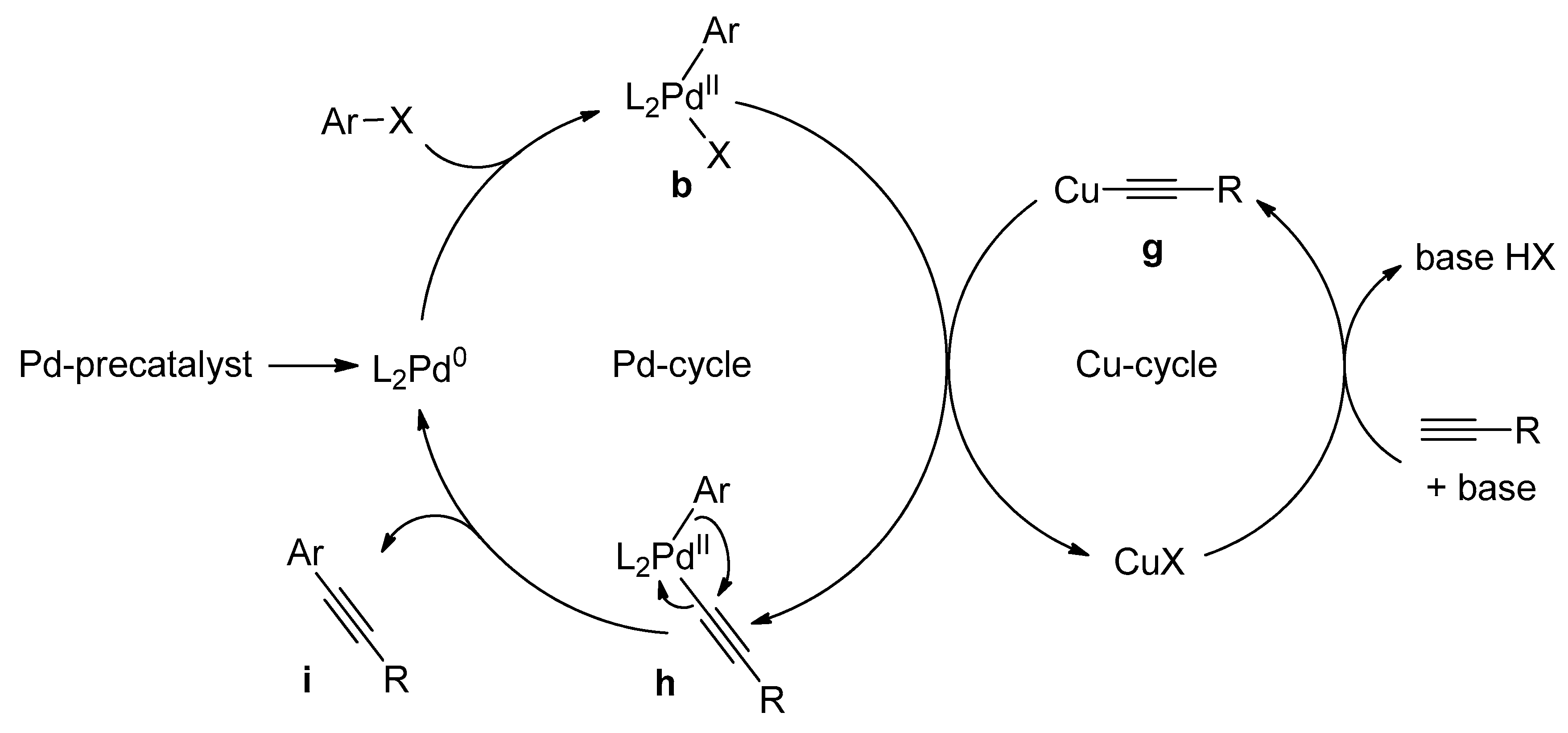
Catalysts | Free Full-Text | Microwave-Assisted Palladium-Catalyzed Cross-Coupling Reactions: Generation of Carbon–Carbon Bond | HTML

Arylation of heterocyclic compounds by benzimidazole-based N-heterocyclic carbene-palladium(II) complexes - ScienceDirect

PDF) Heck reaction using palladium-benzimidazole catalyst: synthesis, characterisation and catalytic activity

Formation of polynuclear palladium complexes with the benzimidazole-2-thiolate anion | Semantic Scholar

PDF) Palladium-Catalyzed Aryl C H Activation and Tandem ortho- Hydroxylation/Alkoxylation of 2-Aryl Benzimidazoles: Cytotoxicity and DNA-Binding Studies | Satish Manda - Academia.edu
![PDF] catena-Poly[[bis(1H-benzimidazole-κN 3)palladium(II)]-μ-benzene-1,4-dicarboxylato-κ2 O 1:O 4] | Semantic Scholar PDF] catena-Poly[[bis(1H-benzimidazole-κN 3)palladium(II)]-μ-benzene-1,4-dicarboxylato-κ2 O 1:O 4] | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/08fac28127f1e880caa63d6e1acc0cab376947a4/1-Table2-1.png)
PDF] catena-Poly[[bis(1H-benzimidazole-κN 3)palladium(II)]-μ-benzene-1,4-dicarboxylato-κ2 O 1:O 4] | Semantic Scholar
![1-Methyl-3-[1-[2-(3-methylbenzimidazol-1-yl)naphthalen-1-yl]naphthalen-2-yl] benzimidazole;palladium(2+);2,2,2-trifluoroacetate | C40H26F6N4O4Pd - PubChem 1-Methyl-3-[1-[2-(3-methylbenzimidazol-1-yl)naphthalen-1-yl]naphthalen-2-yl] benzimidazole;palladium(2+);2,2,2-trifluoroacetate | C40H26F6N4O4Pd - PubChem](https://pubchem.ncbi.nlm.nih.gov/image/imgsrv.fcgi?cid=24874065&t=l)
1-Methyl-3-[1-[2-(3-methylbenzimidazol-1-yl)naphthalen-1-yl]naphthalen-2-yl] benzimidazole;palladium(2+);2,2,2-trifluoroacetate | C40H26F6N4O4Pd - PubChem

PDF) 5-Nitrobenzimidazole containing Pd(II) catalyzed C-C cross-coupling reactions: The effect of the N-substituent of the benzimidazole structure on catalyst activity

Synthesis, crystal structures and biological activities of palladium(II) complexes of benzimidazole and 2-methylbenzimidazole - ScienceDirect

Table 1 from Substituted 2-(2''-pyridyl)benzimidazole palladium(II) complexes as an efficient catalytic system for Suzuki--Miyaura cross-coupling reactions | Semantic Scholar

Palladium(ii) ligated with a selenated (Se, CNHC, N-)-type pincer ligand: an efficient catalyst for Mizoroki-Heck and Suzuki-Miyaura coupling in water. - Org. Biomol. Chem. - X-MOL
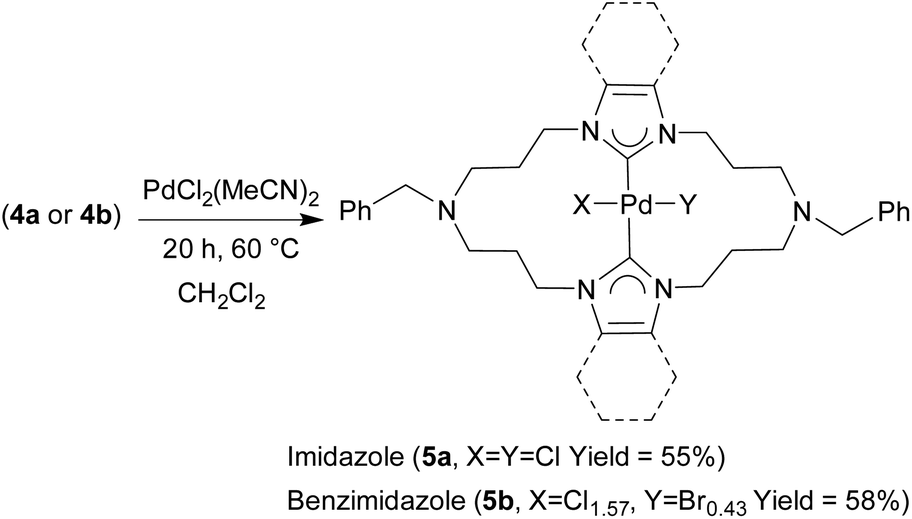
Synthesis and structural characterization of 20-membered macrocyclic rings bearing trans -chelating bis(N-heterocyclic carbene) ligands and the cataly ... - Dalton Transactions (RSC Publishing) DOI:10.1039/C9DT02147G

PDF) Heck reaction using palladium-benzimidazole catalyst: synthesis, characterisation and catalytic activity
![P,N]-phosphinobenzimidazole ligands in palladium-catalyzed C-N cross-coupling reactions: The effect of the N-substituent of the benzimidazole scaffold on catalyst performance - ScienceDirect P,N]-phosphinobenzimidazole ligands in palladium-catalyzed C-N cross-coupling reactions: The effect of the N-substituent of the benzimidazole scaffold on catalyst performance - ScienceDirect](https://ars.els-cdn.com/content/image/1-s2.0-S0022328X17302395-fx1.jpg)
P,N]-phosphinobenzimidazole ligands in palladium-catalyzed C-N cross-coupling reactions: The effect of the N-substituent of the benzimidazole scaffold on catalyst performance - ScienceDirect