Functionalized nitrogen ligands (CN) for palladium catalyzed cross-coupling reactions (part II) - ScienceDirect

Molecular ligand modulation of palladium nanocatalysts for highly efficient and robust heterogeneous oxidation of cyclohexenone to phenol | Science Advances

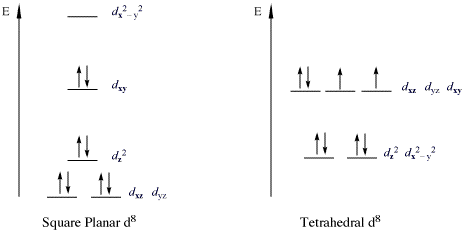
Ligand-enabled C(sp 3 )-H activation a, C-H activation of aliphatic... | Download Scientific Diagram

Cationic Two-Coordinate Complexes of Pd(I) and Pt(I) Have Longer Metal- Ligand Bonds Than Their Neutral Counterparts - ScienceDirect
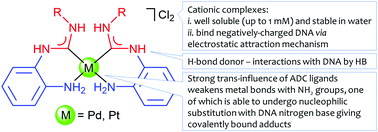
Water soluble palladium(ii) and platinum(ii) acyclic diaminocarbene complexes: solution behavior, DNA binding, and antiproliferative activity - New Journal of Chemistry (RSC Publishing)

A Highly Active Ylide‐Functionalized Phosphine for Palladium‐Catalyzed Aminations of Aryl Chlorides - Weber - 2019 - Angewandte Chemie International Edition - Wiley Online Library

1,2,3-Triazolylidene palladium complex with triazole ligand: Synthesis, characterization and application in Suzuki–Miyaura coupling reaction in water - ScienceDirect

Olefin Dimerization and Isomerization Catalyzed by Pyridylidene Amide Palladium Complexes - Organometallics - X-MOL

Ligand‐Controlled Palladium‐Catalyzed Pyridylation of 1‐tert‐Butoxycarbonyl‐3‐iodoazetidine: Regioselective Synthesis of 2‐ and 3‐Heteroarylazetidines - Zhang - 2017 - Advanced Synthesis & Catalysis - Wiley Online Library
![2,2′-Bipyridin]-6(1H)-one, a Truly Cooperating Ligand in the Palladium-Mediated C–H Activation Step: Experimental Evidence in the Direct C-3 Arylation of Pyridine - J. Am. Chem. Soc. - X-MOL 2,2′-Bipyridin]-6(1H)-one, a Truly Cooperating Ligand in the Palladium-Mediated C–H Activation Step: Experimental Evidence in the Direct C-3 Arylation of Pyridine - J. Am. Chem. Soc. - X-MOL](https://xpic.x-mol.com/paperImg%2F2020_03_12%2FJournal_of_the_American_Chemical_Society_All_img%2F10.1021_jacs.8b10680.gif)
2,2′-Bipyridin]-6(1H)-one, a Truly Cooperating Ligand in the Palladium-Mediated C–H Activation Step: Experimental Evidence in the Direct C-3 Arylation of Pyridine - J. Am. Chem. Soc. - X-MOL
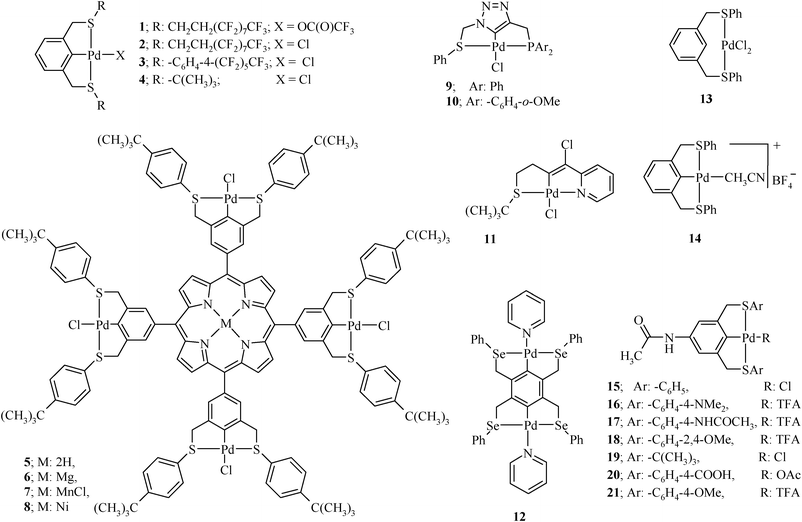
Organochalcogen ligands and their palladium( ii ) complexes: Synthesis to catalytic activity for Heck coupling - RSC Advances (RSC Publishing) DOI:10.1039/C2RA20508D
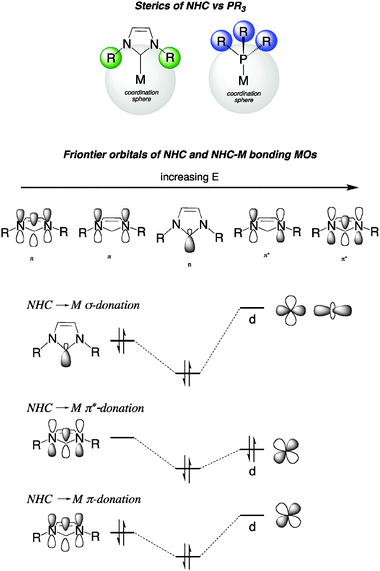
N-Heterocyclic carbene (NHC) ligands and palladium in homogeneous cross-coupling catalysis: a perfect union - Chemical Society Reviews (RSC Publishing)
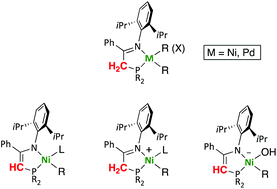
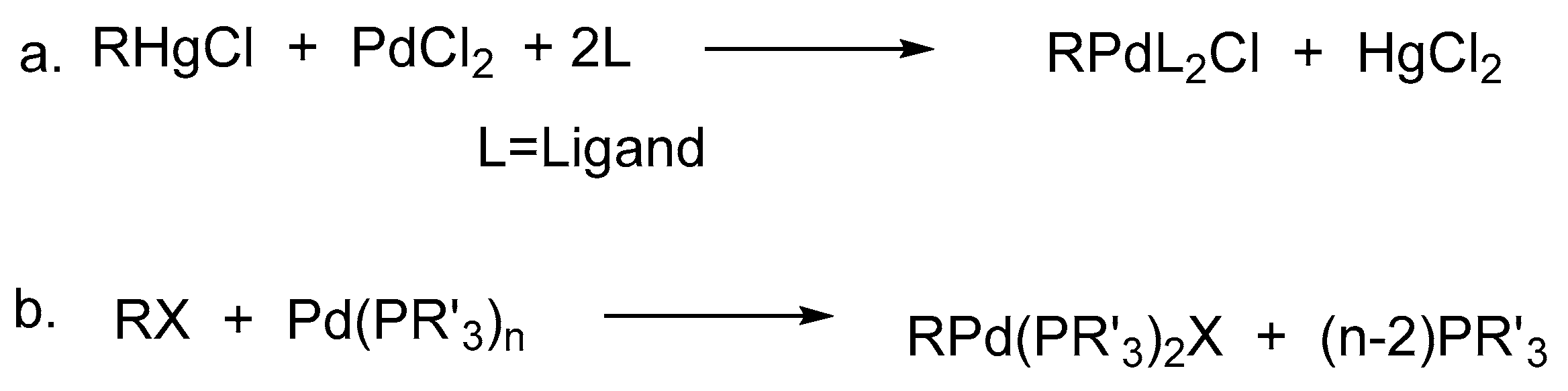
Neutral, cationic and anionic organonickel and -palladium complexes supported by iminophosphine/phosphinoenaminato ligands - Dalton Transactions (RSC Publishing)

Abnormal N-Heterocyclic Carbene–Palladium Complexes for the Copolymerization of Ethylene and Polar Monomers,ACS Catalysis - X-MOL

Ligand‐Assisted Palladium(II)/(IV) Oxidation for sp3 CH Fluorination - Sun - 2016 - Advanced Synthesis & Catalysis - Wiley Online Library

Heterogeneously catalyzed phosphine-free heck cross-coupling reaction of aryl halides with reusable palladium(II) schiff base complex
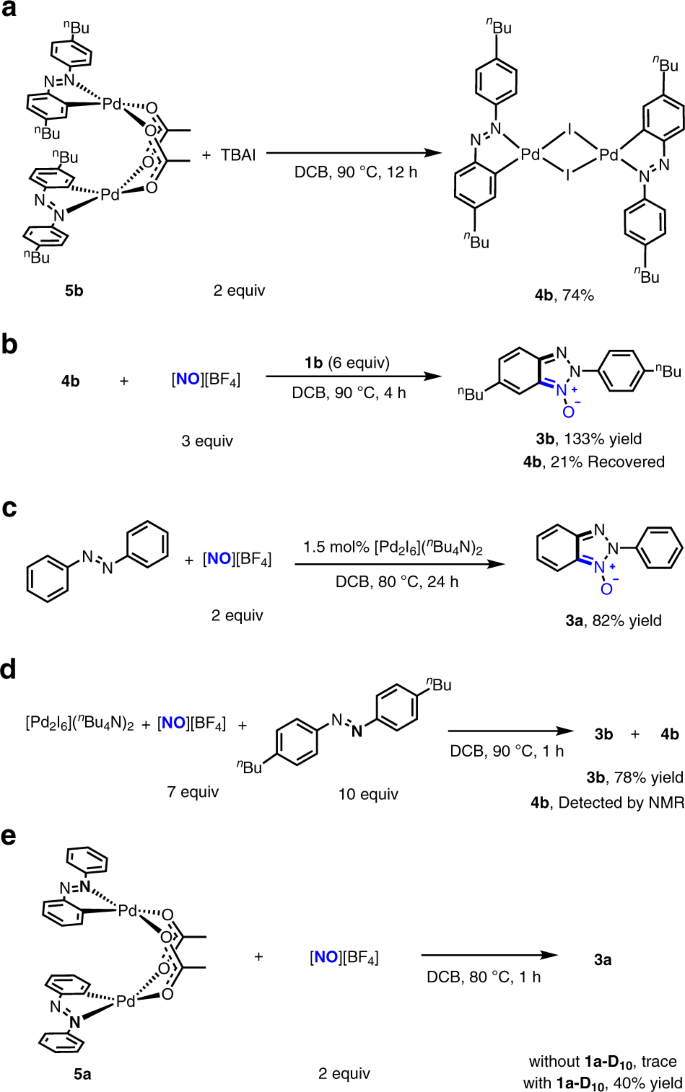
Iodide-enhanced palladium catalysis via formation of iodide-bridged binuclear palladium complex | Communications Chemistry

Axially Chiral Cyclic Diphosphine Ligand-Enabled Palladium-Catalyzed Intramolecular Asymmetric Hydroarylation - ScienceDirect

Design and synthesis of chiral 1,10-phenanthroline ligand, and application in palladium catalyzed asymmetric 1,4-addition reactions - ScienceDirect