Graphene anchored palladium complex as efficient and recyclable catalyst in the Heck cross-coupling reaction - ScienceDirect
![Plot between rate of reaction (-dc/dt) vs [H + ] and [KCl] at T = 303K.... | Download Scientific Diagram Plot between rate of reaction (-dc/dt) vs [H + ] and [KCl] at T = 303K.... | Download Scientific Diagram](https://www.researchgate.net/profile/Ajaya_Singh2/publication/240976537/figure/fig3/AS:669005698318346@1536514796550/Fig-3-Plot-between-rate-of-reaction-dc-dt-vs-H-and-KCl-at-T-303K-NBP.png)
Plot between rate of reaction (-dc/dt) vs [H + ] and [KCl] at T = 303K.... | Download Scientific Diagram
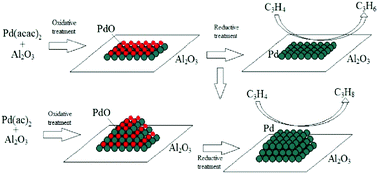
Transformation of the active component during oxidative and reductive activation of the palladium hydrogenation catalyst - New Journal of Chemistry (RSC Publishing)
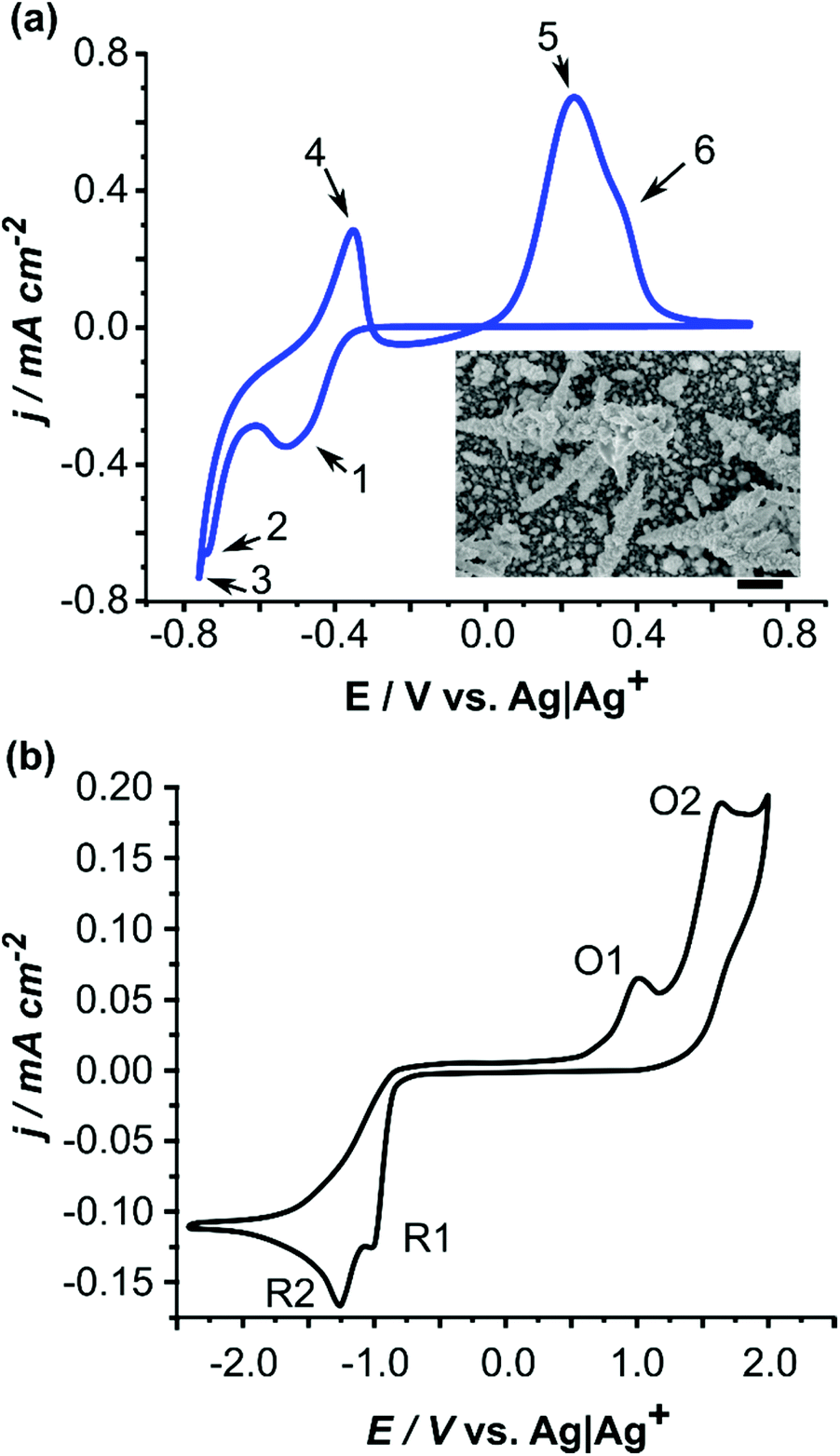
Switching on palladium catalyst electrochemical removal from a palladium acetate–acetonitrile system via trace water addition - Green Chemistry (RSC Publishing)
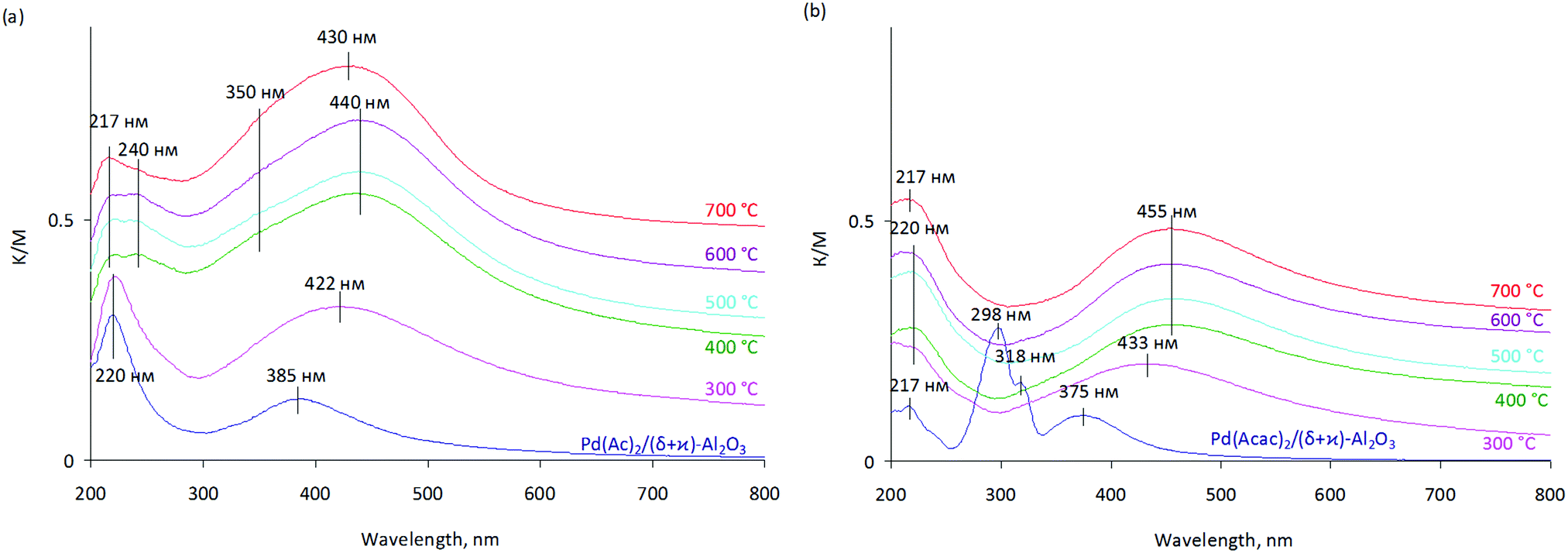
Transformation of the active component during oxidative and reductive activation of the palladium hydrogenation catalyst - New Journal of Chemistry (RSC Publishing)
Spectrophotometric determination of the platinum metals VI.* Determination of rhodium and palladium with 4-(2-thiazolylazo)resor
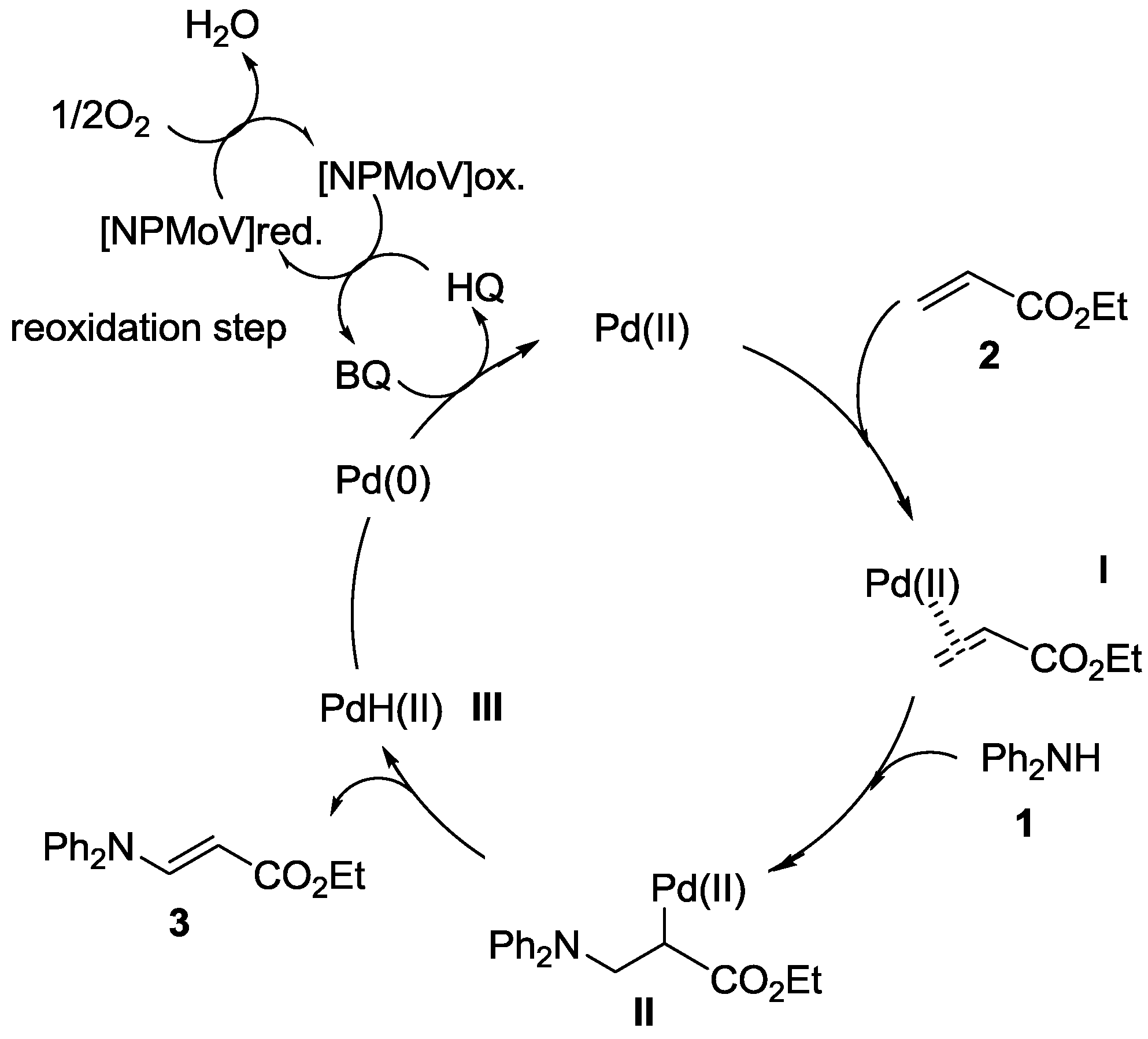
Catalysts | Free Full-Text | Palladium-Catalyzed Intermolecular Oxidative Amination of Alkenes with Amines, Using Molecular Oxygen as Terminal Oxidant | HTML
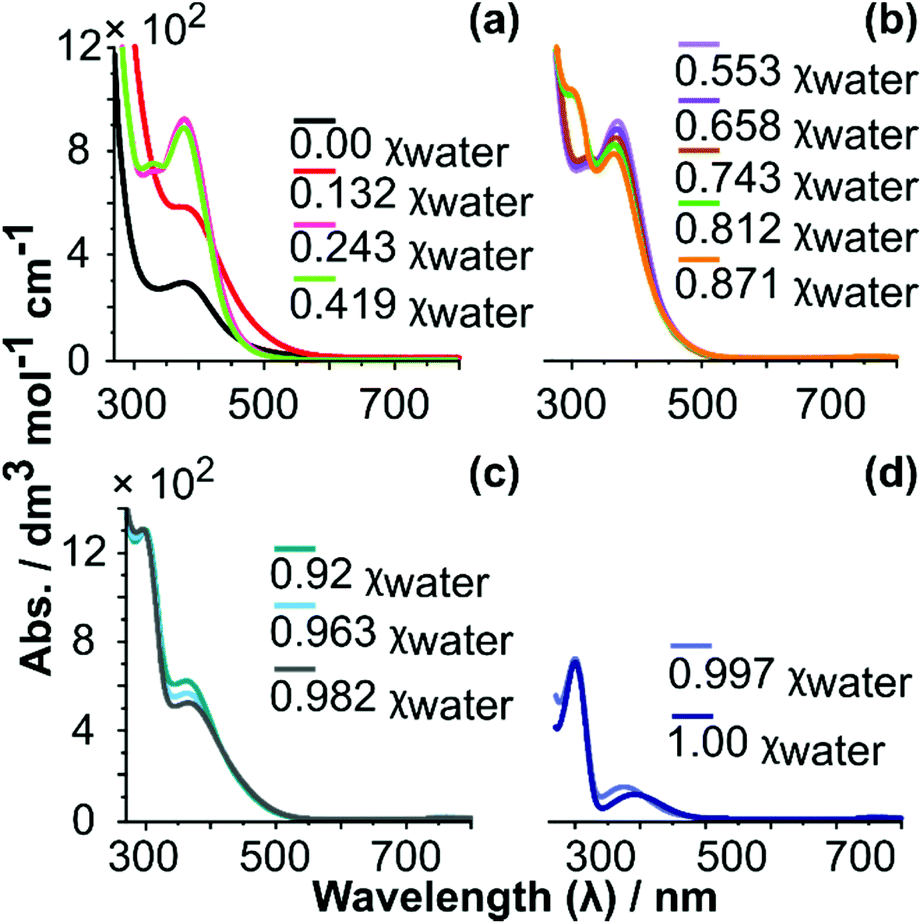
Switching on palladium catalyst electrochemical removal from a palladium acetate–acetonitrile system via trace water addition - Green Chemistry (RSC Publishing)
Synthesis and characterisation of palladium(II) complexes with new diphosphine ligands. Application in the alternating copolymer

Spectra of (a) 0.05 mM Pd(COO(CH 3 )) 2 and (b) 0.01 mg cm − 3 Pd par- | Download Scientific Diagram
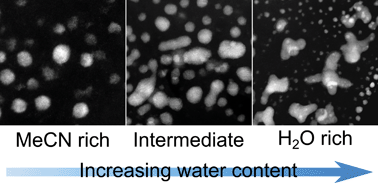
Controlling palladium morphology in electrodeposition from nanoparticles to dendrites via the use of mixed solvents - Nanoscale (RSC Publishing)
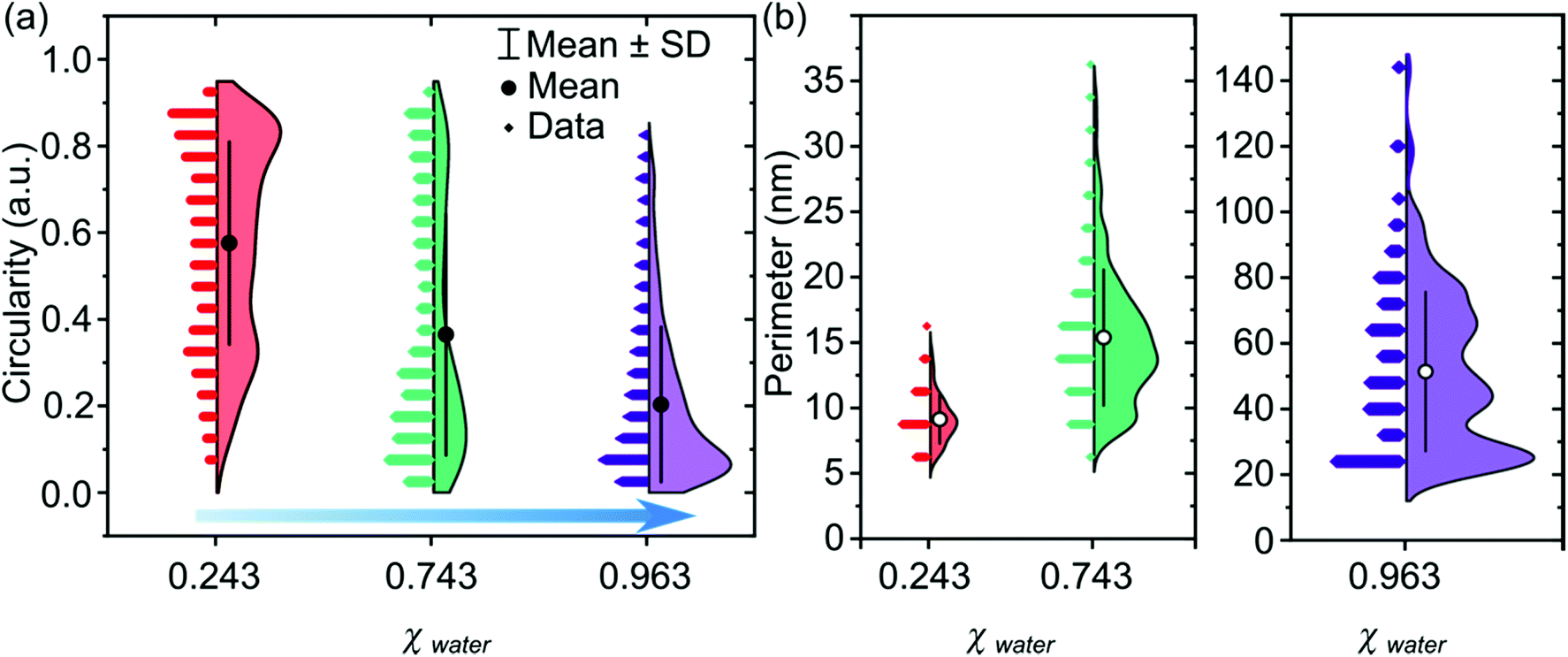
Controlling palladium morphology in electrodeposition from nanoparticles to dendrites via the use of mixed solvents - Nanoscale (RSC Publishing)
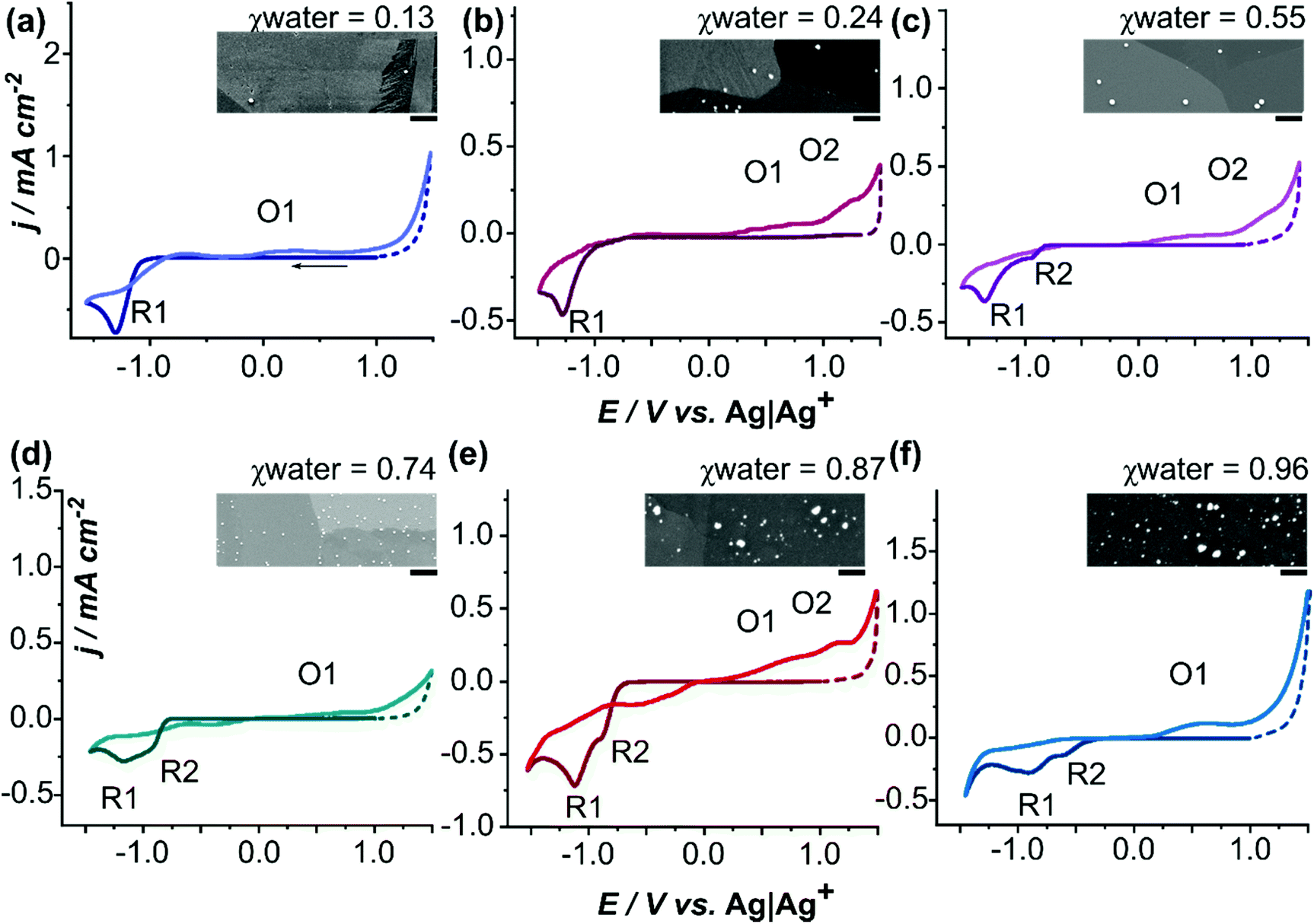
Switching on palladium catalyst electrochemical removal from a palladium acetate–acetonitrile system via trace water addition - Green Chemistry (RSC Publishing)
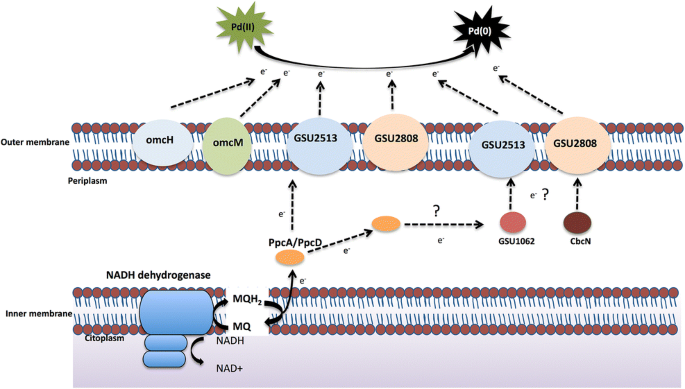
Global transcriptional analysis of Geobacter sulfurreducens under palladium reducing conditions reveals new key cytochromes involved | SpringerLink

Activating electrochemical catalytic activity of bio-palladium by hybridizing with carbon nanotube as “e − Bridge” | Scientific Reports
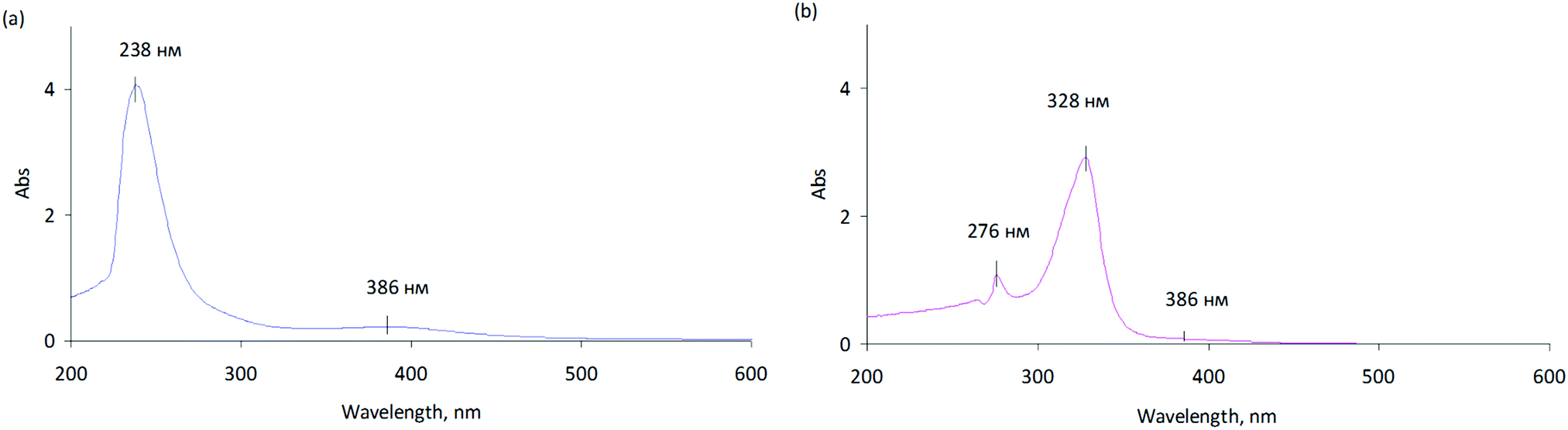
Transformation of the active component during oxidative and reductive activation of the palladium hydrogenation catalyst - New Journal of Chemistry (RSC Publishing)
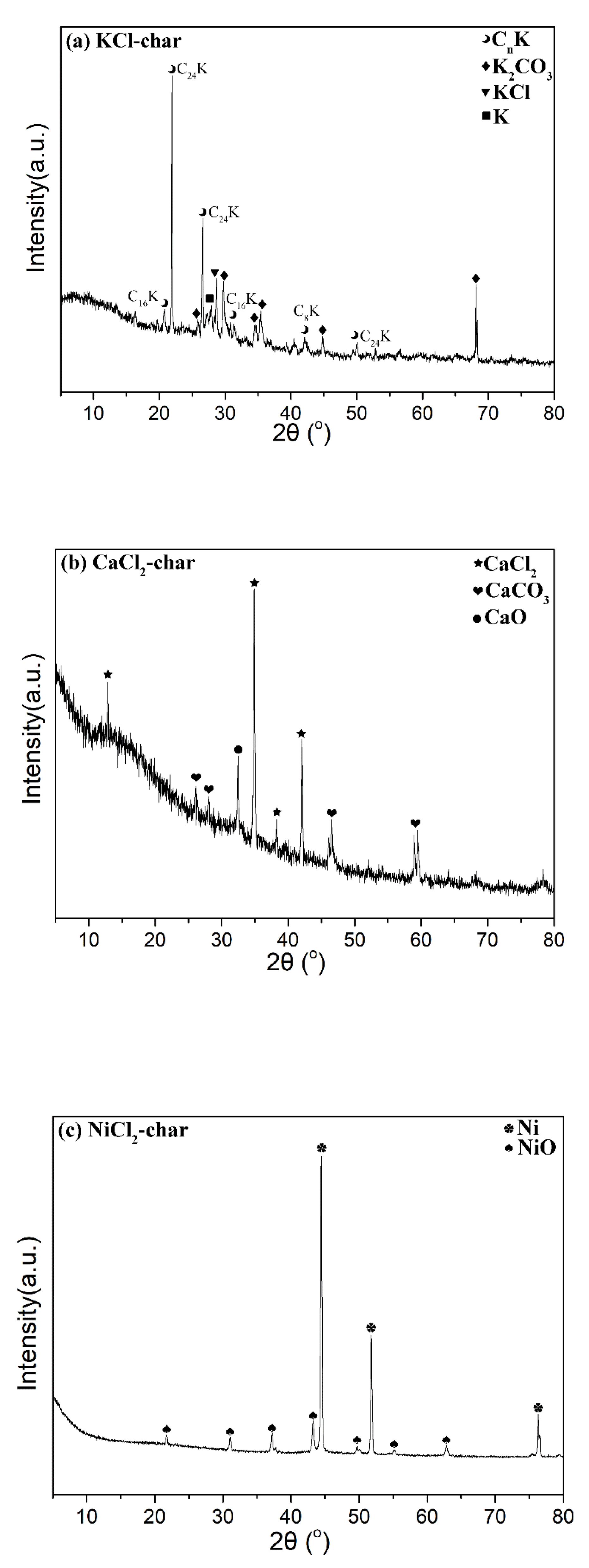
Catalysts | Free Full-Text | Kinetics and Mechanisms of Metal Chlorides Catalysis for Coal Char Gasification with CO2 | HTML

Ligand characteristics and in situ generation of Pd active species towards CC coupling using series of 2-(1H-imidazol-2-yl)phenols - ScienceDirect





![Effect of varying [KNO 3 ] and [KCl] on the rate of oxidation of D-fru... | Download Table Effect of varying [KNO 3 ] and [KCl] on the rate of oxidation of D-fru... | Download Table](https://www.researchgate.net/profile/Yokraj_Katre/publication/275552100/figure/tbl3/AS:669689210474507@1536677758457/Effect-of-varying-KNO-3-and-KCl-on-the-rate-of-oxidation-of-D-fru-at-303-K.png)